Anaesthetic drug Target Control Infusion
The goal of this task is to develop a test-bench platform for an autonomous infusion pump controller for Depth of Anaesthesia (DoA) and to use the testbench to validate the proposed technologies.
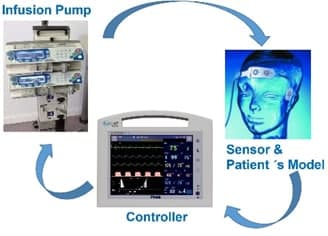
An intelligent infusion controller for Vital Signs is a medical device that regulates the specific vital signs parameter such as Blood Pressure (BP), Neuromuscular transmission (NMT) or Depth of Anaesthesia (DoA). For example, with a BP controller, the system operates delivering vasoactive drugs; the ultimate goal is to reduce the patient´s hypertension, and precisely control blood pressure measurements in a patient undergoing surgical intervention in OR (Operating Room) or in postcardiac surgery in ICU. (Intensive Care Unit). In the Use Case, we plan to integrate partners’ technical contribution and demonstrate one round of continuous engineering utilizing FOCETA technologies, which starts from modelling, semantic reasoning, simulation, automatic test case generation, to abnormality detection, cyber-security defence, automatic repair (including LEC incremental training) and redeployment.
Step 1: Baseline model
A prototypical implementation of the testbench and autonomous intelligent controller will be implemented. This baseline implementation will be amenable to continuous improvement based on the semantic modelling/reasoning techniques. It includes a monitor and controller, with external OEM infusion pump tree; a target depth of anaesthesia level profile is set up for the operation. The target control infusion SW component computes at regular intervals the new drug dose (with e.g. propofol) to be infused using machine learning techniques. The patient´s model determines the Depth of anaesthesia (DoA) level achieved. The patient is modelled using traditional compartmental analysis in which the interpersonal variability is considered. This DoA value is transferred via a serial channel to the infusion controller. The main focus will be on validation and verification of the HiL (Hardware -in-the-loop) system in virtual test platforms with the tool landscape for this purpose provided by FOCETA. HiL tests will be performed in laboratory, with the concept of continuous improvement in mind. In parallel, a Virtual Platform for the autonomous infusion pump controller will be built, to explore the different RNN control strategies and testing its autonomous mode.
Step 2 – Execute the system and generate virtual test cases
Subsequently, we plan to execute the system. During operation time, different targets are set up, and in each test case, different patient´s models are used, considering weight, gender, pathology, etc. The created traces are collected and new variations out of the trace are collected in order to “challenge” the system. This set up will mimick as much as possible the real life conditions, so that performance, safety and security are considered. For predicting the patient´s behaviour a mixed approach will be used, combining advantages of data-based and model-based techniques using the data-based and model-based methods; mining formal specifications from the observed time series, to learn the rules defining the nominal operation of the system. The test platform will be created and validated in the test laboratory developed based on the scenario definition and formal specification.
Step 3 – Detect abnormal situations that are not considered previously
It will be ensured that the behaviour works according to its specification that incudes safety requirements. The automated system is complemented by runtime monitoring and runtime enforcement development. The infusion controller in the simulated environment is equipped with advanced runtime monitors to detect potential abnormal behaviours. (e.g. the patient´s sensitivity to the drug is modified, and thus the patient´s model is altered, etc). The virtual platform will be used to verify the safety requirement for the pump controller and to explore the different RNN control strategies and testing its autonomous mode. The technology of non-intrusive debugging will be used for detecting the anomaly.
Step 4 – Improve the unit by considering the abnormal situation
Once an abnormal behaviour is detected, the automated controller should switch to supervisory mode to allow updates in an online or offline fashion. To improve the unit considering the abnormal situation, two types of actions can take place:
- Stage 1: Modify the control strategy (e.g. with RNN), and make a new control plan proposal with patient´s behaviour forecasting, for the anaesthesiologist to approve.
- Stage 2: Make operation autonomous, without anaesthesiologist intervention. Run testing techniques to examine if the decision is biased towards some pre-listed criterion.
